| [_menu/menu.htm] |
Reforming Electrolytic
Capacitors
Manufacturers claim that most old
electrolytics can be saved if the correct procedure is followed,
regardless of how long they have been unused. Such capacitors must be
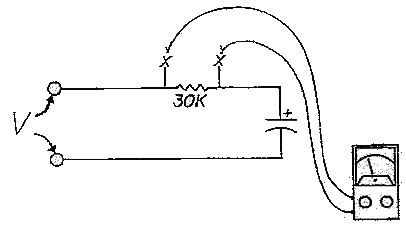
"reformed". This process consists of applying rated voltage through a
resistance (about 30,000 ohms, five watt) for five minutes plus one minute
for each month of storage (see figure 6). As the capacitor reforms, the
voltage across the resistor will drop (measured at the Xs in Figure 6). If
that voltage will not drop below 10% of applied voltage after one hour,
the capacitor is probably beyond help.
Reforming Electrolytic Capacitors
The process of
reforming an old aluminum electrolytic capacitor consists of the
application of rated voltage, through a resistor, for a period equal to
five minutes plus one minute per month of storage.
The
electrolytics appearing on the surplus market have often been in storage
for a very long period indeed. Some manufacturers use a visible code, of
which the first two digits indicate the year of manufacture.
The
circuit shown in the sketch above works reasonably well. Apply the rated
voltage through a 5W resistor. Anything from 20K-50K will do, as this is
far from a precision process. The meter is used to measure the voltage
drop across the resistor; when no current is flowing, there will be no
drop. Obviously, when there is a large voltage drop (more than 20% of the
applied voltage), there must be a significant current flow through the
capacitor. The nature of a proper capacitor is to impede DC current flow,
so when there is such flow, something must be wrong.
Note: Apply
the appropriate D.C. voltage to the capacitor with a D.C. power supply. An
old Kepco, Lamba etc. tube regulated lab power supply rig works great. Be
sure to observe the proper polarity!
Within an aluminum
electrolytic there is a large area of aluminum foil and an electrolytic
paste. As the voltage is applied, current flows until aluminum oxide forms
on the surface of the foil, because aluminum oxide is a very good
insulator. If excess voltage has been applied during the electrolytics
lifetime, it is possible that tiny welds exist which the oxide insulator
cannot separate. When that occurs, the capacitor cannot "reform", and
should be discarded.
If the amount of current flow (voltage drop
across the resistor) is great initially, that is not a problem. If it
doesn't start dropping within five minutes of application of voltage, a
definite hazard exists. The current flow indicated that energy is being
dissipated within the capacitor, in the form of heat. Excess heat may
"pop" the electrolytic, causing the paste to spit out...a threat to eyes
and paint.
It's also worth remembering (one forgets only once) that
a good capacitor will store its energy for quite a while, and discharge it
through the hand when picked up. It's smart, then, to discharge the unit
deliberately, through a resistor equal to about one ohm per volt of
charge.
A new capacitor should rapidly take a charge right to rated
voltage, in which case only a small voltage drop will appear across the
resistor. It is possible to reform capacitors in the circuit, of course,
but if rectification is by solid state diodes and there is a large current
flow, it is possible to destroy one or more of the diodes, or to damage
the transformer.
Electrolytic capacitors can be dangerous. They can
be charged to a high voltage and will retain that energy for quite a
while. If the terminals of associated circuitry are touched, a severe
shock and burn may result.
Another hazard associated with
electrolytics is "spitting". Each of these cans is filled with, among
other things, a thick fluid which can be extremely irritating. A small
rubber safety plug is fitted to most electrolytics of recent manufacture.
When the capacitor fails, internal pressure may go too high; the plug will
blow and the fluid will spit out.
Electrolytic capacitors of a
given capacity and voltage will vary considerably in configuration and
size, from one manufacturer to the another. Ideally, there will be chassis
space to permit mounting the "twist-lock" variety. Otherwise, the tubulars
(such as the Sprague TVL 1720) must be packed, glued or clipped wherever
space is available.
NOTE: The preceding information was obtained
from an old copy of Tu-Be Or Not Tu-Be Modification Manual by H.I.
Eisenson.
The Electrolytic Capacitor
The origin of the
electrolytic capacitor or condenser can be traced to the second half of
the 19th century when the discovery was made that film can be formed on
aluminum electrochemically and that it will exhitbit unidirectional
electrical conductance and other peculiar properties. One of the early
attemped applications of the electroltyic capacitor was in conjunction
with the starting of single-phase induction motors; efforts were also made
to utilize it for power factor correction in alternating current circuits.
It appears that no extensive use had been made of this device until the
early twenties, when its utility in filter circuits supplying rectified
plate current to radio tubes was definitely establisehd. These capacitors
were"polarized" and of the "wet" type. Several years later "dry"
electroltyic condensers of a low voltage rating and large capacitance
found a limited application in A-battery eliminators (comprising a
rectifier and a filter circuit) which furnished filament current for d.c.
radio tubes. By 1929 the high-voltage dry electrolytic was developed and
soon found very extensive and diversified applications in several
fields. The annual production of dry and wet electrolytic capacitors
amounts now to tens of millions and they are used in radio receivers and
transmitters, sound systems and other electronic apparatus, in telephone
circuits, in conjuction with electric motors and, to a smaller extent, in
a number of other applications.
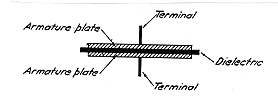
The capacitor may be considered as
a device for storing static electricity. The essential parts of a
capacitor are two electrodes, which consist of conducting members, closely
spaced by a dielectric or insulating medium. The electrodes are usually
metallic plates or foils, while the dielectric may be vacuum, gases (for
instance, air), liquid (like mineral or vegetable oil), or solids (like
mica, glass, wax-impregnated paper, and so on).
The capacitance of a condenser is a measure of the
quantity of electricity that can be stored in it at a given potential
(voltage). The unit of capacitance is the farad and it corresponds to a
charge of one columb at one volt pressure across the terminals of the
device. These relations are expressed by the formulae: Q=CE, C+Q divided
by E or where C=farads, E=volts, Q=columbs.
As the farad is too
great a unit for practical purposes, the microfarad (MF)-one millionth of
a farad-or its subdivisions are ordinarily used. The energy in joules,
stored in a capacitor, equals one half CE squared.
The capacitance
of a condenser is directly proportional to the area of the electrodes
spaced by the dielectric and is inversely proportional to the thickness of
the latter. The nature of the dielectric is the third determining factor
for the capacitance. If this dielectric medium is a mineral oil, the
capacitance of the condenser may be, for instance, twice as great as it
would be with air, everything else being the same. With castor oil the
capacitance will be about five times as great as with air. The ratio of
the capacitance of a condenser with a given dielectric medium between the
electrodes to the capacitance of the same condenser with air (or, more
accurately, vacuum) as a dielectric is designated the dielectric constant
of the medium or its specific inductive capacitance.
Electroltyic
capacitors consitute one of the several classes of capacitors. To
differentiate between the electroltyic and all other classes we shall
designate the latter in this text as "nonelectroltyic".
The
structure of the electroltyic capacitor comprises the fundamentally
important component parts present in any capacitor-the electrodes and the
dielectric between them. It also performs the characteristic function of
storing and releasing electrostatic charges. However, the electroltyic
capacitor possesses in addition some very distinct structural and
functional features which justify placing it in a class of its
own.
The essential difference between the electrolytic and
nonelectroltyic capacitor resides in the nature and thickness of the
respective dielectrics and in the presence or absence of an ionic
conduction medium (the electrolyte) between the metallic electrodes. In
the nonelectroltyic capacitor the thickness of the dielectric is usually
not less than the gauge of a thin sheet of paper, while in the
electroltyic capacitor the dielectric is many times thinner. In the former
class of capacitors the dielectric is made of materials of well-known
compositions, like mica, wax- or oil-impregnated paper, glass, oil and the
like, while in the latter class the true nature of the effective
dielectric has not been definitely established. We know, however, that the
dielectric in electrolytic capacitors is intimately associated with
surface of the electrode and that its existence is correlated with the
formation of an oxide film on the latter.
Another important
characteristic of the electroltyic capacitor is the marked ionic
conductivity of the medium positioned between the metallic electrodes, in
contrast to the highly insulating material (mica, oil, and the like)
between the electrodes in nonelectroltyic capacitors.
From the
point of view of usage, it is typical of the electroltyic capacitor that
it combines some remarkably valuable advantages for...(certain
specific)...applications. The most outstanding advantage of the
electroltyic condenser resides in the great, in some cases even enormous,
capacitance per unit of the electrode area which it exhibits at moderate
(100-600V), but particularly at low voltages (down to a few volts).
The following example may illustrate the great compactness of the
low-voltage electroltyic capacitor as compared with an equivalent
wax-paper capacitor. A unif of the former class designed for use in a
unidirectional circuit and rated for 2,000 MF at 5 to 10 volts (depending
on the ripple voltage) can be housed in a container of about 10 cubic
inches, while the capacitance of the lowest voltage condenser of the
latter class, occupying the same space, will be of the order of only a few
MFs. Consequently, the bulk, weight and also the cost of the electroltyic
capacitor are in this case less to a very great degree.
The
cause of this impressive difference between the two kinds of capacitors
will be discussed in the following paragraphs. As already stated, for a
given type of dielectric, the less its thickness the greater the
capacitance per unit electrode area. The limit for increasing the
capacitance by this expedient in nonelectroltyic condensers is determined
by the gauage of the thinnest available insulators which will provide
the required dielectric strength. For example, it is difficult to make
condenser paper thinner than 0.0003", and it is the usual practice in the
manufacture of wax- or oil-impregnated paper capacitors to place at least
two layers of paper between the foils, to prevent breakdowns due to
unavoidable pinholes and conducting particles (metallic specks, and the
like) in the paper. Thus, 0.0006" between the elctrodes appears to be the
minimum spacing. But even if a chance were taken with a single layer of
paper between the foils of a condenser for very low voltages, the ultimate
minimum spacing would be 0.0003", which would also set the limit to the
capacitance per unit area for this type of dielectric, no matter how low
the voltage rating of the unit. Thus, whether the latter were intended for
5 volts or 25 volts, it could not occupy a space less than that required
for the housing of a section of the required capacitance wound with a
0.0003" paper betwen the foils.
With electroltyic capacitors, however, such limitations do not exist as
the minimum thickness of the dielectric is not determined by the gauge of
the layer or spacer placed between the electrodes. The almost intangible
dielectric of the electroltyic capacitor is formed electrochemically on
the electrode surface, and the accurate control of its thickness, down to
less than one millionth of an inch, can be readily effected. Thus,
by the simple expedient of varying the filming voltage, which governs
the thickness of the dielectric, one may produce condensers, within
wide voltage and capacitance ranges, which can be housed in containers of
the same dimensions. For example, a container of 10 cubic inches will be
suitable for the assembly of a capacitor rated for 100 MF and formed at
100V or of a capacitor of 400 MF at 25V. Hence, the electroltyic capacitor
exhibits a remarkable adapabiliy to the operating voltage.
Finally, the unilateral properties of the film (or its conductance in
one direction only) make it possible to use electrolyitc capacitors for
the blocking of direct current in the undesired direction. The unilateral
feature makes the electroltyic capacitor also more adaptable to circuits
in which unidirectional pulsating currents are flowing; in such cases, by
making the condenser polarized or asymmetric in its action, its bulk and
cost can be substantially halved without reducing its capacitance.
Contrasting with this, nonelectroltyic condensers are always symmetric and
no analogous economy is feasible when they are used on pulsating current,
nor can they be employed to block or restrict the flow of d.c. in one
direction only.
Most of the electroltyic capacitors are of the
polarized or asymmetric type and can be used only if the voltage impressed
across their terminals is unidirectional. Furthermore, they must be
connected with the proper polarity, or damage to the capacitor and
associated apparatus may result. The unidirectional feature, however, may
be turned into an advantage in special cases as has pointed out in the
forgoing. The electroltyic capacitor can even be made semipolarized to
block the flow of d.c. in one direction and to restrict it to a
predetermined value in the opposite direction.
Nonelectroltyic capacitor sections or units can be built for much
higher operating voltages than is practicable for the electrolytic type.
Furthermore, the nonelectrolytic capacitor can be operated continuously on
alternating current at its rated value.
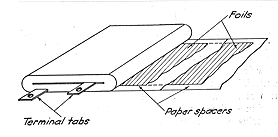
The conventional dry
electroltyic capacitor comprises a winding very similar in its appearance
to that of a wax- or oil- paper nonelectrolytic capacitor. The winding
consists of two foils of which at least one must be of a film-forming
metal; in commercial electroltyic capacitors both foils are practically
without exception of aluminum. They are interleaved with paper layers
impregnated in a suitable electrolyte. The latter is known as nonaqueous,
as it contains not more than a few percent of water. As a rule, no
unabsorbed electrolyte is present in the container and it is due to this
fact that these spacer-wound capacitors are designated as "dry",
regardless of the fluidity or solidity of the impregnant.
Since the foils are very closely spaced, usually between 0.0001" and
0.0006", the resistivity of the electrolyte may be comparatively high
without unduly increasing the resistance of the path for the current
flow.
Because of the close spacing of the electrode foils and the
thin gauge of the latter (down to 0.0005"), the dry capacitor can be
built very compactly, saving space and weight. Furthermore, two or even
more sections of the same or of a widely different voltage ratings can be
assembled in one container, without interference among the sections, as
the latter can be effectively insulated from one another.
The
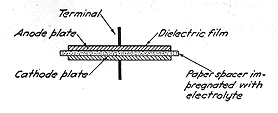
Electrodes of the Electroltyic Capacitor
The true cathode of the electrolytic capacitor is the electrolyte (or,
more accurately, its ions, which thus play the part of one of the
electrodes), the film is the dielectric and the anode is the second
electrode represented by the metallic member on the surface of which the
film is formed. However, to establish a good contact between the
electrolyte and the external circuit, a second metallic member is
required; the latter is in intimate contact with the electroltye and is
called in practice the cathode, although it serves primarily to distribute
the current over, or pick it up from, the electrolyte. This arrangement is
characteristic of polarized or asymmetric capacitors, intended for
operation with unidirectional potentials. For such use a single dielectric
film is sufficient and the second electrode-the cathode-does not have to
be necessarily of a film-forming metal. It is essential, however, to
select for its construction a material which is not attacked by the
electrolyte and which will not contaminate the latter. It must, of course,
meet the usual strucrual requirements and be of moderate cost. The cathode
foils of dry electroltyic capacitors are made of aluminum, though of a
lower grade than required for the anodes, a purity of about 99% being
satisfactory. Aluminum has been preferred for cathode foil because of its
comparatively low price, light weight and consequently great coverage,
ease of fabrication and winding. It is not apt to corrode while in storage
and is not attacked by the usual electrolytes, nor does it contaminate
them.
In nonpolarized or symmetric capacitors, used in a.c. circuits, the
electrolyte is again the true cathode, but in this case two aluminum
electrodes, both provided with dielectric films, must be used. Each of the
films, however, is fully effective only while it is subjected to a positve
potential, i.e. during the respective positive half cycles. On the
negative half cycles the dielectric properties of the films are
alternately reduced to a small value and during that time the foils on
which they are formed serve to distribute the current over the
electrolyte by conduction.. The arrangement in this capacitor is
equilvalent to the series-oppositon connection of two polarized units.
With this combination the two units become alternately effective and
substanially ineffective as the polarity is reversed. In both analogous
cases, as the voltage alternates, the electric charges are being shifted
from the one film to the other back and forth. It must be noted, however,
that the symmetric electrolytic capacitor differs basically from the
series combination of two nonelectroltyic capacitors. In the lattter case
both dielectrics remain fully effective at all times and the electric
charges in the two units rise and fall simultaneously.
The above
material was taken largely from THE ELECTROLYTIC CAPACITOR by Georgiev,
1945. |



